 
Each element has a unique set of energy levels, and thus the transition from higher to lower energy levels produces X-rays with frequencies that are characteristic to each element. Outer-shell electrons then fall into the inner shell, emitting quantized photons with an energy level equivalent to the energy difference between the higher and lower states. 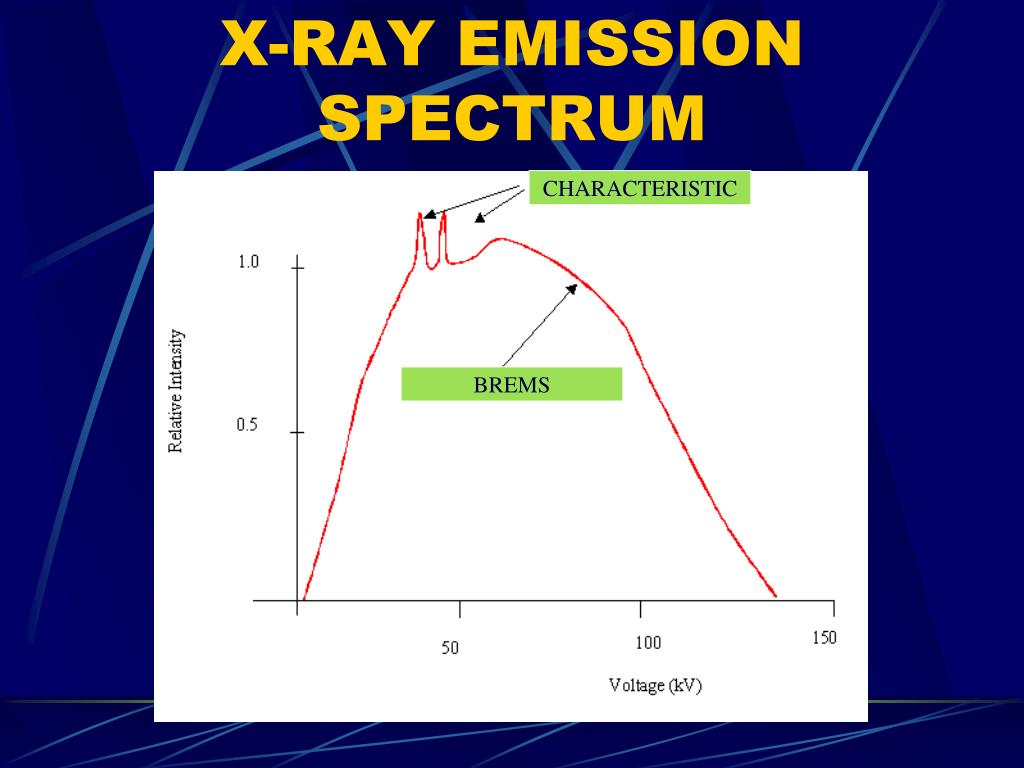
After the electron has been ejected, the atom is left with a vacant energy level, also known as a core hole. When the incident particle strikes a bound electron (the target electron) in an atom, the target electron is ejected from the inner shell of the atom. Characteristic X-rays were discovered by Charles Glover Barkla in 1909, who later won the Nobel Prize in Physics for his discovery in 1917.Ĭharacteristic X-rays are produced when an element is bombarded with high-energy particles, which can be photons, electrons or ions (such as protons). 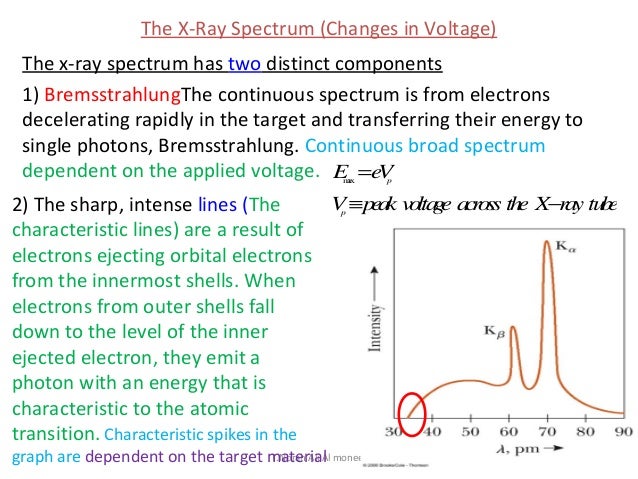
Characteristic X-rays are emitted when outer- shell electrons fill a vacancy in the inner shell of an atom, releasing X-rays in a pattern that is "characteristic" to each element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
